March 31, 2025: Senator asks how FDA will be accountable for unregulated active pharmaceutical ingredient (API) imports
Major Stories
Senator Jim Banks of Indiana has asked the U.S. Food and Drug Administration how it will prevent unvetted shipments of the active pharmaceutical ingredients (API) in injectable weight loss drugs like Ozempic from entering the country. Federal law requires that API shipments come from FDA-registered facilities and that they are accompanied by certificates of analysis, but PSM’s research into semaglutide and tirzepatide imports shows that many unlawful shipments are still making it into the U.S.
Patient safety issues in the GLP-1 space this week
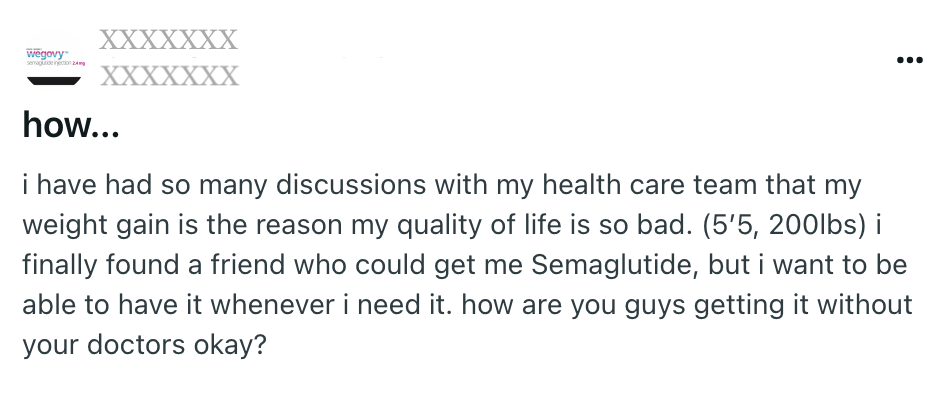
Post found on Reddit, March 2025
GLP-1s are revolutionary medicines that can make a huge difference in the lives of diabetics and people suffering from obesity, but they come with significant side effects and must be prescribed by a doctor. “Getting it without your doctor’s okay” can be very dangerous.
Share this handout to explain the problem with unapproved APIs.
Domestic News
Ohio’s pharmacy board shut down a spa for fake Botox. States continue to consider PBM reform.
The Ohio Board of Pharmacy suspended the license of a beauty spa after determining that it was injecting patients with counterfeit Botox it had acquired from an unnamed dentist in California. This action comes after the board suspended the licenses of two companies on charges they were treating patients with unapproved weight-loss drugs in January.
Texas pharmacists rallied in Austin to urge lawmakers to support bills that would prevent pharmacy benefit managers (PBM) from under-reimbursing them. The Alabama legislature is considering a bill that would require PBMs to reimburse community pharmacies at the state’s Medicaid rate, which includes a $10.64 dispensing fee that would help cover the cost of operating a pharmacy. The states are among several considering PBM reform to protect independent pharmacies like this drugstore in Bath, Maine, which is closing after almost 110 years, in part because low reimbursements from PBMs and insurance companies don’t cover service costs.
The Obesity Action Coalition and The Obesity Society sent a letter urging the FDA to enforce federal regulations around compounding following the recent resolution of GLP-1 medicine shortages.
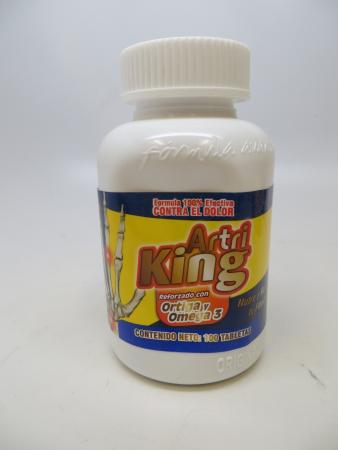
The FDA warned about Artri King in 2022.
International News
Pill press news in Canada and the United Kingdom. Fentanyl precursors allegedly coming from a company in India.
Royal Canadian Mounted Police in Quebec shut down two fake pill operations, seizing three pill presses, more than a kilogram of suspected methamphetamine and over 200,000 pills, including 8,000 counterfeit Xanax pills.
Members of a dark web drug trafficking ring that sold as many as 11 million counterfeit Xanax pills made in garden sheds and garages in England's West Midlands were cumulatively sentenced to more than 40 years in prison.
A U.S. federal court has charged an India-based chemical company and three high-level employees with illegally importing fentanyl precursor chemicals. A March 2025 indictment alleges that the defendants worked with an undercover agent to arrange the sale of more than 6,000 kilograms of precursors to the U.S. and Mexico between March and November 2024.
In its Annual Threat Assessment the Office of the Director of National Intelligence noted that while China remains the primary source for fentanyl precursors and pill presses, drug trafficking organizations in Mexico are also sourcing precursor chemicals from India.
Libya’s Ministry of Health warned hospitals and medical centers not to distribute counterfeit versions of cancer treatment called Lonsurf.