News
Prescription Drug Freight Fraud Report, April 2026
What do FDA records of GLP-1 and antibiotic imports show about freight fraud patterns over the last six months?
[...]What’s master carton smuggling?
In March, customs officers in Cincinnati announced the seizure of thousands of shipments of GLP-1s and other peptides packaged inside more than 300 “master cartons” with deceptive manifests meant to slip into the country unnoticed. Curious how this smuggling scheme works? Read on.
[...]April 20, 2026: Maryland becomes the second state to set a drug price cap
The Jardiance upper payment limit, based on the Medicare maximum fair price, is set to take effect in 2027.
[...]April 13, 2026: New video shows Mochi Health-affiliated pharmacy’s “corner cutting”
A newly-released March 2025 Washington State Department of Health video shows investigators raiding Aequita, where “cutting corners [was seen] at nearly every level of its operation.”
[...]The FDA speaks up about AFPs and drug importation
Is personal drug importation legal in the context of alternative funding programs? The FDA says no.
Are self-funded health plans allowed to import medicine because of “Section 804” or “Florida’s importation plan”? Also, no.
[...]April 6, 2026: Over 200 patients bought unapproved imported peptides from a now-indicted Utah physician
The doctor allegedly bought the unapproved drugs through a middleman and relabeled them before selling them to patients.
[...]March 30, 2026: A second pharmacy benefit manager settles suit with the FTC over insulin price inflation
CVS Health’s Caremark deal mirrors the deal the FTC struck with Express Scripts last month, a person familiar with the case told Reuters.
[...]March 24, 2026: Physician sentenced for administering expired injectables in long-running fraud scheme
A federal judge in Alaska sentenced rheumatologist Dr. Claribel Tan to six-and-a-half years in prison for administering substandard drugs to patients while fraudulently billing insurers
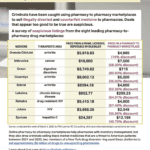
[...]Using pharmacy-to-pharmacy marketplaces? Read this Suspicious Sales Surveillance Report
Online pharmacy-to-pharmacy marketplaces help with inventory management, but they also require vigilance because they carry a higher risk of being exploited by criminals selling black market medicines.
[...]Counterfeit drug crimes typically earn light sentences. How did Safe Chain’s co-owners, the Boyd brothers, end up with 18 and 20 years?
Wire fraud charges drove much higher penalties than the shipping of unsafe medicine into the supply chain, and the Boyds’ sentences were based on the government proving high-dollar, deliberate fraud.
[...]