Article Topic
PSM written testimony regarding Michigan Senate Bills 3, 4, and 5
PSM submits written testimony on Michigan Senate Bills 3, 4, and 5 On April 23, The Partnership for Safe Medicines submitted written testimony in opposition to Michigan Senate Bills 3, 4, and 5, which would create a prescription drug affordability board (PDAB) in Michigan to establish upper payment limits on certain medicines. Read the testimony…
[...]Prescription Drug Affordability Board Activity through April 16, 2025
Prescription Drug Affordability Board Activity through April 16, 2025 Activities Summary Colorado: Colorado’s PDAB met on April 11, 2025, where it addressed an error in All-Payer Claims Database data and how it affected affordability review information for Enbrel, Cosentyx and Stelara. Maryland: Maryland’s PDAB did not meet in April. Oregon: Oregon’s PDAB met on April…
[...]April 21, 2025: Kentucky doctor pleads guilty after injecting patients with semaglutide from unregistered sources
A Kentucky doctor treated weight loss patients with research chemicals; CBP seized 90,000 alprazolam pills being smuggled into the U.S.
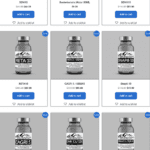
[...]FDA Alert: Counterfeit Ozempic found in U.S. Supply Chain
The FDA and Novo Nordisk are warning the public about counterfeit Ozempic injections circulating in the U.S. drug supply chain. The falsified products, labeled with lot number PAR0362 and serial numbers beginning with 51746517, were seized by the FDA on April 9, 2025. Their contents and safety are unverified and pose serious health risks.
[...]Prescription Drug Affordability Board Activity through March 31, 2025
Prescription Drug Affordability Board Activity through March 31, 2025 Activities Summary Colorado: Colorado’s PDAB cancelled its March meetings. Maryland: Maryland’s PDAB met on March 24, 2025 to begin a cost review of Farxiga and review feedback about updates to PDAB regulations. Oregon: Board members met on March 19, 2025, where they elected a subset of…
[...]April 14, 2025: Regulators announce a third instance of counterfeit Ozempic injections in the U.S. drug supply
The FDA announced that it had seized counterfeit Ozempic injections on April 9. PSM testified at a congressional hearing.
[...]April 7, 2025: A study examines FDA-OCI’s enforcement actions over a five-year period
A study examined the 130 enforcement actions undertaken by the U.S. Food and Drug Administration’s Office of Criminal Investigations from 2016 through 2021.
[...]FOIA Returns from towns using Alternative Funding Vendors
PSM has sent open records requests to several towns and schools with self-funded health plans about their relationships with Alternative Funding Vendors.
[...]March 31, 2025: Senator asks how FDA will be accountable for unregulated active pharmaceutical ingredient (API) imports
Senator Jim Banks asked pointed questions about how FDA will stem the tide of semaglutide and tirzepatide coming from unknown facilities.
[...]Government Sentencing Memo, USA v Aaron Michael Thomas, February 24, 2025
United States District Court Northern District of Oklahoma USA v Aaron Michael Thomas Government Sentencing Memo Filed February 2025 Read the document.
[...]