Alert
ED-Marketed Dietary Supplement Recalled Due to Presence of Prescription Drugs
This recall has been initiated after an FDA laboratory analysis found the product to contain undeclared sildenafil and/or tadalafil. Sildenafil and tadalafil are ingredients in FDA approved products for the treatment of male erectile dysfunction in the family of drugs known as phosphodiesterase (PDE-5) inhibitors. The presence of sildenafil and/or tadalafil in Adam’s Secret Extra Strength products makes them unapproved new drugs for which the safety and efficacy have not been established and therefore subject to recall.
[...]FDA Puts All Mexico-Produced Hand Sanitizers on Import Alert
Under the import alert, alcohol-based hand sanitizers from Mexico offered for import are subject to heightened FDA scrutiny, and FDA staff may detain the shipment. As part of their entry review, FDA staff will consider any specific evidence offered by importers or manufacturers that the hand sanitizers were manufactured according to U.S. current good manufacturing practice requirements. This marks the first time the FDA has issued a countrywide import alert for any category of drug product.
[...]Methanol-Contaminated Rubbing Alcohol Recalled by FDA
This is a reprint of an FDA Alert. Essaar Inc. Issues Voluntary Nationwide Recall of Rubbing Alcohol Contaminated with Methanol When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company’s announcement as a public service. FDA does not endorse either the product or the company. Company Name: Essaar Inc.…
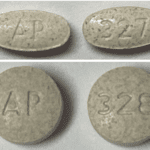
[...]FDA Alert: Acella Pharmaceuticals, LLC Issues Voluntary Nationwide Recall of Two Lots of NP Thyroid®, Thyroid Tablets, USP Due to Sub Potency
Risk Statement: Patients being treated for hypothyroidism (underactive thyroid), who receive sub potent NP Thyroid®, may experience signs and symptoms of hypothyroidism (underactive thyroid) which may include, fatigue, increased sensitivity to cold, constipation, dry skin, puffy face, hair loss, slow heart rate, depression, swelling of the thyroid gland and/or unexplained weight gain or difficulty losing weight. There is reasonable risk of serious injury in newborn infants or pregnant women with hypothyroidism including early miscarriage, fetal hyperthyroidism, and/or impairments to fetal neural and skeletal development. In elderly patients and patients with underlying cardiac disease toxic cardiac manifestations of hyperthyroidism may occur, such as cardiac pain, palpitations or cardiac arrhythmia. To date, Acella has received four reports of adverse events for these lot numbers possibly related to this recall.
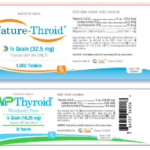
[...]RLC Labs, Inc., Issues Voluntary Nationwide Recall of All Lots of Nature-Throid® and WP Thyroid® with Current Expiry Due to Sub Potency
Risk Statement: Patients being treated for hypothyroidism (underactive thyroid), who receive sub potent Nature-Throid® or WP Thyroid®, may experience signs and symptoms of hypothyroidism (underactive thyroid) which may include, fatigue, increased sensitivity to cold, constipation, dry skin, puffy face, hair loss, slow heart rate, depression, swelling of the thyroid gland and/or unexplained weight gain or difficulty losing weight.
[...]CorgioMed LLC Issues Voluntary Nationwide Recall of All Lots of Leafree Instant Hand Sanitizer Aloe Vera Labeled as EDIBLE ALCOHOL
Risk Statement: Ingesting hand sanitizer, which is intended for topical use, may result in alcohol toxicity. Symptoms of alcohol toxicity may range from lack of coordination, slowed or slurred speech, drowsiness to coma, which can be fatal. Young children may experience a sharp decrease in blood sugar which may result in death. Pregnant women who ingest alcohol have experienced birth defects and developmental disabilities. Nursing mothers who ingest alcohol in above moderate levels may see developmental, growth and sleep pattern damages in their babies and may experience impaired judgement and ability to safely care for their child.
[...]FDA Alert: Food and Drug Administration Issues a More Serious Warning About Methanol in Hand Sanitizers
The FDA has issued a second, more serious warning about hand sanitizers that have been made with deadly methanol. Their first warning came on July 2, when they warned they had “seen a sharp increase in hand sanitizer products that are labeled to contain ethanol (also known as ethyl alcohol) but that have tested positive for methanol contamination.”
[...]AAA Cosmética, S.A. de C.V. Issues Voluntary Nationwide Recall of all Lots Within Expiry of bio aaa Advance Hand Sanitizer 480 mL Due to the Potential Presence of Undeclared Methanol (Wood Alcohol)
AAA is recalling all lots of their hand sanitizer due to the presence of undeclared methanol (wood alcohol) in their hand sanitizer products.
[...]4e Brands North America Issues Nationwide Voluntary Recall of Hand Sanitizer Due to Potential Presence of Undeclared Methanol (Wood Alcohol)
4e Brands of San Antonio, Texas is recalling all lots of their hand sanitizer due to the presence of undeclared methanol (wood alcohol) in their hand sanitizer products.
[...]FDA Alert: MasterPharm, LLC. Issues Voluntary Nationwide Recall of Finasteride Plus 1.25mg Due to the Presence of an Undeclared Antihypertensive Drug
This is a reprint of an FDA Alert. When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company’s announcement as a public service. FDA does not endorse either the product or the company. May 11, 2020 Company Contact Information Consumers: Lin Leung, MasterPharm, LLC Phone: (866) 630-5600 Email: recall@masterpharm.com…
[...]