Importation
Drug importation is not a new idea.
Drug Importation Is Not A New Idea, And It Is Not A Good One, Either–More Than A Decade Of Experience Has Shown It Is Dangerous
[...]CNN Warns that Fake Drugs are a Global Industry That Puts Your Life at Risk
In a Mosaic Science article shared by CNN, author Srinath Perur delves into the global pharmaceutical industry and discovers that in India, counterfeit and substandard medications are an everyday occurrence.
[...]PSM’s letter asking the President and Congress to protect Americans from dangerous, imported medicines
Letter Asking the President And Congress to Protect Americans from Imported Drugs, October 30, 2018 Today, The Partnership for Safe Medicines (PSM) sent a letter to President and Members of Congress urging them to protect Americans from the threat of unregulated, imported medicines flowing into our borders. October 30, 2018 Dear President Donald J.…
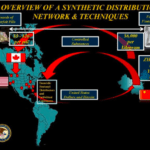
[...]Drug Importation and the Deadly Challenge of Screening 275 Million Packages a Year
The Food and Drug Administration (FDA) in their report U.S. Food and Drug Administration and the International Mail Facilities (IMFs), describes the daunting job that Customs and Border Protection (CBP) faces when attempting to weed out counterfeit medications and packages containing illicit fentanyl. In 2017, IMFs received 275 million packages. Of these, 10,000 were screened by CBP, and of those 86% contained drugs. The investigation of a suspect package is incredibly time-consuming; an experienced FDA investigator might take as long as 20 minutes to process a package containing just on product.
[...]Congress Passes Opioid Epidemic Legislative Package
The Partnership for Safe Medicines applauds the passage of the SUPPORT Act, an $8 billion package which will help develop non-addictive painkillers, improve prescription drug monitoring programs, establish comprehensive opioid recovery centers, and strengthen Customs and Border Protection’s ability to intercept fentanyl that is illegally shipped into the United States.
[...]Pharmacist and Drug Safety Advocate Urge HHS to Consider the Dangers of Importation
In this October 16, 2018 for Drug Topics, Alliance of Safe Online Pharmacies advisor Libby Baney and pharmacist and American Pharmacists Association CEO Thomas Menighan discuss the risks to public safety posed by drug importation.
[...]Importation Has Some Huge Risks
There are certainly things we can learn from other countries’ healthcare successes, but importation is not the right approach.
[...]Discussing the Difficulty of the Import Problem
This distressing situation shows that deadly fentanyl analogues are killing Americans who are using laced non-opioid prescriptions.
[...]Canadian Law Enforcement Veteran Urges Cross-Border Cooperation to Fight Fentanyl and Other Counterfeit Medicines
In this August 21, 2018 editorial for the Havre Daily News Canada Border Services Agency and the Ontario Provincial Police veteran Don Bell talks about the need for Canada and the United States to work together to combat counterfeit drugs.
[...]National Sheriffs’ Association Sends Letter of Concern About Importation of Prescription Drugs
On August 8, 2018, the National Sheriffs’ Association wrote the President to oppose prescription drug importation. Importing drugs from other countries, they say, “jeopardizes law enforcement’s ability to protect the public health and endangers the safety of law enforcement and other first responders.”
[...]