Posts Tagged ‘FDA Alert’
FDA Alert: Class 1 Recall of Some Mesa Biotech COVID Tests Due to Risk of False Positives/Contamination
This is a reprint of an FDA Alert. Mesa Biotech, Inc., Recalls Certain Accula SARS-CoV-2 Tests for Risk of False Positives Caused by Contamination The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death. Recalled Product Product Name: Accula…
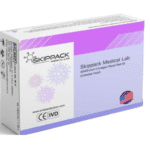
[...]FDA Alert: Class 1 Recall on Skippack Medical Lab SARS-CoV-2 Antigen Rapid Test
This is a reprint of an FDA Alert. Do Not Use Skippack Medical Lab SARS-CoV-2 Antigen Rapid Test: FDA Safety Communication May 10, 2022 The U.S. Food and Drug Administration (FDA) is warning people not to use the Skippack Medical Lab SARS-CoV-2 Antigen Rapid Test (Colloidal Gold). This test is not authorized, cleared, or approved…
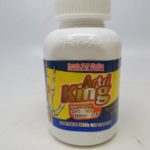
[...]FDA Alert: Herbal Arthritis Treatments Contain Dangerous Prescription Drugs
FDA has received adverse event reports, including of liver toxicity and death, associated with the use of Artri King products, since the agency issued its first warning about an Artri Ajo King product on January 5, 2022.
[...]FDA Alert: Class 1 Recall of Unauthorized Covid-19 Tests
This is a reprint of an FDA Alert. SD Biosensor Recalls STANDARD Q COVID-19 Ag Home Tests That Are Not Authorized, Cleared, or Approved by the FDA and May Give False Results The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious…
[...]Dietary Supplement Sold on Amazon Recalled for Containing Prescription Drug Ingredients
Esupplementsales, LLC was informed by Amazon that laboratory analysis has found the product to be tainted with tadalafil, an ingredient known as a phosphodiesterase (PDE-5) inhibitor found in an FDA approved product for the treatment of male erectile dysfunction. The presence of tadalafil in Hard Dawn Rise and Shine Capsules renders the product an unapproved drug for which safety and efficacy have not been established and, therefore, subject to recall.
[...]California Boutique Recalls Diet Supplements For Containing Banned Sibutramine
Je Dois L’avoir Boutique is voluntarily recalling all of the 365 Skinny High Intensity Pills and or 365 Skinny Emergency Boutique, 30 day capsules supply to the retail/consumer level. The 365 Skinny High Intensity Pills and 365 Skinny Emergency Boutique have been found to contain Sibutramine which is a controlled substance by the DEA.
[...]FDA Alert: Third Dietary Supplement Sold on Amazon Recalled for Containing Prescription Drug Ingredients
On December 17, 2020, FDA issued a press release that warned consumers to avoid certain products found on Amazon, eBay and other retailers due to hidden and potentially dangerous drug ingredients. It also encouraged online marketplaces to ensure these products are not sold on their platforms.
[...]FDA Alert: Second Dietary Supplement Recalled for Containing Prescription Drug Ingredients
On December 17, 2020, FDA issued a press release that warned consumers to avoid certain products found on Amazon, eBay and other retailers due to hidden and potentially dangerous drug ingredients. It also encouraged online marketplaces to ensure these products are not sold on their platforms.
[...]FDA Alert: Dietary Supplement Recalled for Containing Prescription Drug Ingredients
Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using these drug products.
[...]ED-Marketed Dietary Supplement Recalled Due to Presence of Prescription Drugs
This recall has been initiated after an FDA laboratory analysis found the product to contain undeclared sildenafil and/or tadalafil. Sildenafil and tadalafil are ingredients in FDA approved products for the treatment of male erectile dysfunction in the family of drugs known as phosphodiesterase (PDE-5) inhibitors. The presence of sildenafil and/or tadalafil in Adam’s Secret Extra Strength products makes them unapproved new drugs for which the safety and efficacy have not been established and therefore subject to recall.
[...]