federal judge in Florida sentenced Charles and Patrick Boyd to a cumulative 38 years in prison. The brothers owned Safe Chain Solutions, a Cambridge, Maryland-based drug distributor at the center of a diversion ring that endangered the health of American patients by selling U.S. pharmacies over 85,00 counterfeit bottles of secondhand HIV medicine worth more than $250 million.
Sanjay Kumar got 43 months for selling counterfeit Keytruda. PSM wrote federal agencies about a Super Bowl ad promoting diagnostic tests and compounded medicines with false claims.
On February 26, 2026, PSM was glad to see the Indiana legislature pass SB 282, a bill that strengthens compounding and medical spa regulation to keep Indiana patients safer.
As Europol seized a huge amounts of counterfeit pharmaceuticals abroad, a Kentucky legislator proposed a bill to regulate med spas after a constituent was seriously injured and adverse events related to weight loss drugs see surges across the US.
Washington State investigators found serious violations at a Mochi Health-affiliated compounding pharmacy producing GLP-1s, and a med spa bill in Indiana hears public testimony about a med spa bill.
Industry experts told us that these two companies are on the FDA’s Green List, despite problematic inspections in late 2024 and early 2025. Learn what the inspectors found.
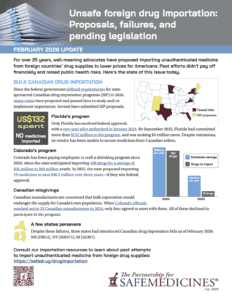
Well-meaning advocates continue to propose importing medicine from foreign countries to lower drug prices for Americans. Past efforts didn’t pay off financially and raised public health risks. Here’s the state of this issue today.
Our podcast covers the latest in pharma crime and medicine safety.
Like your information on video? Subscribe to our YouTube playlist!

Congressman Krishnamoorthy is looking into GLP-1 drugs that may be entering the US unlawfully.

The FDA cited a medspa for DSCSA violations. Learn more.

A recent FDA warning letter highlights upstream supply chain vulnerabilities.
Click the images below to see more recent videos.