Search:
Filter:
Sort:
Ozempic, the second drug that the state has set an upper payment limit on, will be capped at $274 for a 30-day supply.
Gilead Sciences has filed a complaint against Pain Relief Rx and other companies that it alleges sold diverted medicines on pharmacy to pharmacy marketplaces.
The ProRx warning letter signals increased FDA scrutiny of compounded weight loss product that are not intended to be compounded in the US.
Criminal counterfeiters are exploiting Americans’ demand for blockbuster diabetes and weight loss drugs made with semaglutide and tirzepatide, while compounders, telehealth companies, and med spas strain and skirt public health measures to meet high demand.
An Arkansas newspaper printed an ad hawking foreign drugs. Are foreign “pharmacies” advertising in your local newspaper? On May 18, 2026, PSM sent a letter rebuking the Arkansas Democrat-Gazette for selling ad space to a Canadian online pharmacy last month. The advertisement, which promised to “save buyer’s up to 80% on their prescription drugs,” promotes the purchase of non-FDA-approved, foreign…
In a major settlement, Made-in-China.com will stop selling research chemicals to U.S. buyers following an investigation by Connecticut’s Attorney General.
The FDA proposed restricting three GLP-1 drugs from the 503B bulks list, which would prevent outsourcing facilities from compounding them from raw ingredients without a clear clinical need.
The Partnership for Safe Medicines strongly supports the Food and Drug Administration’s proposal to exclude semaglutide, tirzepatide, and liraglutide from the 503B bulks list. This action reflects sound science, sound law, and a clear-eyed commitment to patient safety. Full statement inside.
The former doctor admitted to buying tirzepatide from a lab in China and distributing it without medical evaluations, and is now being sued by a former hospital employee who alleges the drug caused life-threatening complications requiring major surgery.
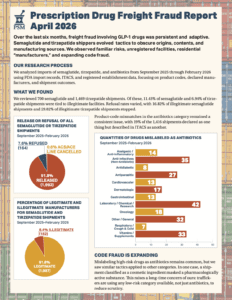
What do FDA records of GLP-1 and antibiotic imports show about freight fraud patterns over the last six months?