Search
Washington (July 19, 2018) – Shabbir Imber Safdar, executive director of the Partnership for Safe Medicines, released the following statement regarding today’s announcement by the Department of Health and Human Services to create a drug importation working group at FDA: “We are deeply concerned about today’s announcement, particularly given the deaths of Americans in at least 26 states over the…
Read MoreThe U.S. Department of Justice announced another guilty plea from the second co-owner of a dietary supplement company based out of Corpus Christi, Texas. Vanessa Gonzales admitted to purchasing pills produced in China that contained undeclared or banned pharmaceutical ingredients…
Read MoreFor the last 15 years, the FDA and HHS have opposed drug importation for safety reasons, but there is another question that is often overlooked: If a pharmacy inadvertently distributes a counterfeit drug it legally purchased from a foreign wholesaler, can the pharmacist be held liable? A 2004 lawsuit, Fagan v. AmerisourceBergen Co, raises disquieting questions.
Read MoreThe U.S. DOJ announced that Nathan Ott of Chambersburg, PA received a 210-month prison sentence. Ott pleaded guilty to purchasing fentanyl by the kilogram online and using it to manufacture counterfeit pills which he and his six co-defendants then sold online and around town…
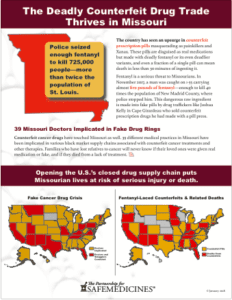
Read MoreKelly Gant is a young woman in Missouri overcoming an addiction to what she thought was Xanax. After being hospitalized for an overdose, she was shocked to learn that she did not overdose on alprazolam, but on fentanyl. It was then that she began to learn more about the bags filled with hundreds of pills that her dealer sold to high school students…
Read More